FDA Authorizes Coronavirus Vaccines for Children Younger than 5
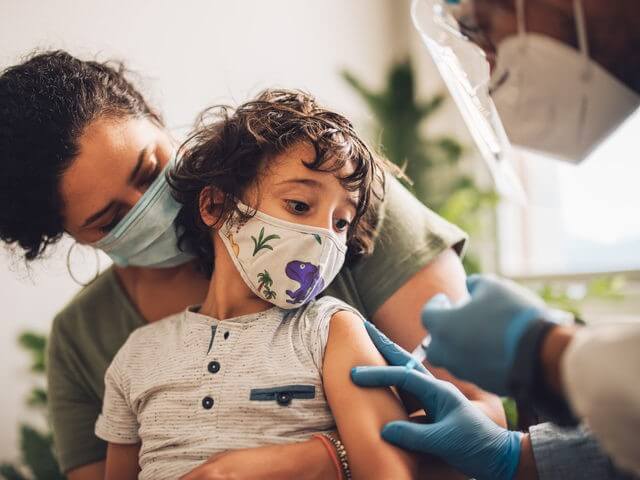
The Food and Drug Administration (FDA) on Friday authorized both Moderna’s and Pfizer’s coronavirus shots for children under the age of five.
“Many parents, caregivers and clinicians have been waiting for a vaccine for younger children and this action,” FDA Commissioner Robert Califf said in a statement Friday.
“As we have seen with older age groups, we expect that the vaccines for younger children will provide protection from the most severe outcomes of COVID-19, such as hospitalization and death,” he added:
The move comes two days after the FDA’s Vaccine and Related Biological Products Advisory Committee unanimously approved the vaccines for young children. The vaccines are now pending final approval from the U.S. Centers for Disease Control and Prevention (CDC). – READ MORE





Responses